RESILIENCE Research Placement Case Study
Hear from Anna Richardson, a natural sciences student studying biology and chemistry at the University of Nottingham. After completing a mini placement with Dr Bernice Wright at UCL and RESILIENCE, she now wants to go into research and development (R&D) investigating medicine or pharmacy.
Observing the operational differences between a professional lab environment and my university teaching lab was very interesting. Following my second year at university, I will be working at Astra Zeneca for a placement year, in one of their R&D teams (doing work similar to that performed during my mini lab placement with Bernice). Bernice was so kind to give me a chance to come into her lab and have a go at investigating mammalian cell culture in a working laboratory - I am so grateful for the amazing experience I had over my days in her lab!

I decided to work on a summer lab placement with UCL Biochemical Engineering to gain experience in a lab outside the University of Nottingham, to not only help me with my future in industry, but because I am extremely passionate about healthcare sciences and allied research. It has been so useful to see the practical application of the techniques I have heard about in lectures and to understand the value of the knowledge I have gained from my Natural Sciences degree. I am excited to move forward with my degree training using my new skills and knowledge.

The aim of my experiment was to investigate the efficacy of LY294002 (an anti-cancer agent/drug) for reducing the viability of HEK293 cells (tumour cells) cultured under artificial 2D and physiological 3D conditions to develop an in vitro testing platform that can be translated to in vivo studies. The experimental design involved culturing HEK293 cells (1×104 cells seeded per experimental condition) on a 2D tissue culture plastic (96 well plate) as well as on 3D polystyrene cell microcarriers. The cells were treated with increasing concentrations (0.1 – 10000 µM) of LY294002, which is a specific inhibitor of phosphoinositide-3-kinase (PI3K), an important regulator of cell proliferation.
Key methods I used to achieve my study objectives were 2D and 3D mammalian cell culture (growing cells in nutrient-rich media in vitro in the presence of LY294002 for 24h). In 2D culture, the cells grew in a single layer in multi-well plates and in 3D culture, the cells grew on the surface of microcarriers. The Alamar Blue assay (measures cell metabolic activity by detecting the reduction of resazurin (low fluorescence) to resorufin (high fluorescence)) so this assay can be used to quantify live cells. Resazurin is coloured blue and as the density of cells increases, the intensity of fluorescence increases because it is reduced to resorufin. The colour changes from blue to purple to pink as cell numbers increase.

Generating results in the laboratory
I used a cell standard curve to derive numbers of metabolically active cells in drug (LY294002) treated samples (Figure 1.). I used a live-dead assay that distinguishes live and dead cells using fluorescent dyes. This assay involves the use of calcein AM (non-fluorescent dye) that enters through the membrane of live cells and is hydrolysed by intracellular esterases into calcein (fluorescent green dye) which is retained by the cell. Ethidium homodimer (fluorescent red dye) enters dead cells through their compromised cell membrane.
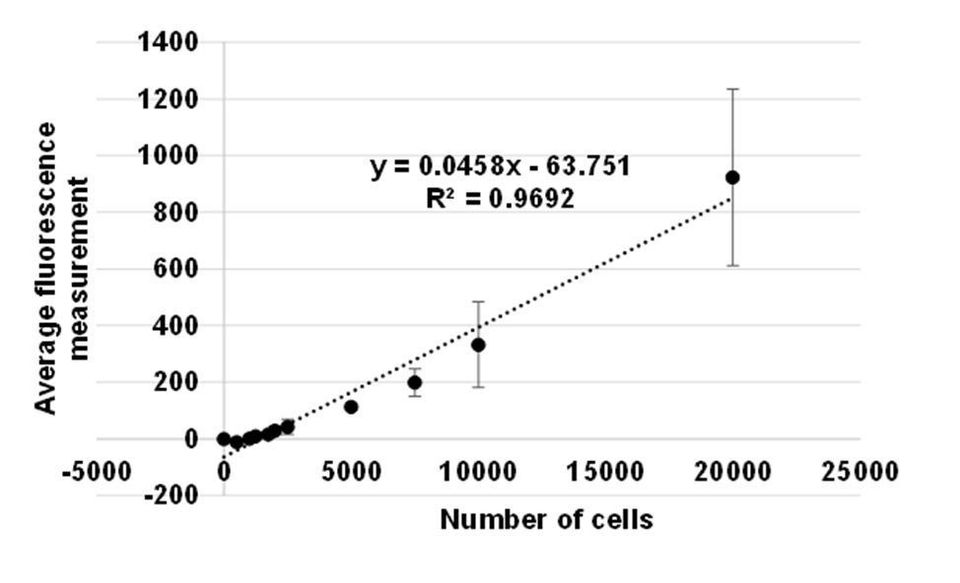
Figure 1. Standard curve correlating HEK293 cell numbers to fluorescence measurement. The scatter plot shows that the average amount of fluorescence (y-axis) increased as numbers of HEK293 cells (x-axis) increased. Variation between technical replicates (n=3) is calculated as standard deviation shown as error bars.
My results showed that HEK293 cells on 3D microcarriers proliferated to a lesser extent than on 2D surfaces (Figure 2.). The number of cells in each experimental condition was 1×104 cells, but less than 1×104 live cells were measured in cell microcarrier controls (untreated and drug vehicle (1% (v/v) dimethylsulphoxide (DMSO)) treated cells), and 1.8×104 – 2.4×104 cells were measured in 2D condition controls. These data indicate that cells did not proliferate under 3D conditions but they proliferated under 2D conditions. LY294002 inhibited cell proliferation under 3D and 2D culture conditions in a dose dependent manner (Figure 2.). Jiang et al. (2010) also demonstrated that LY294002 significantly reduced cell proliferation in human nasopharyngeal carcinoma (CNE-2Z) cells in a dose dependent manner.
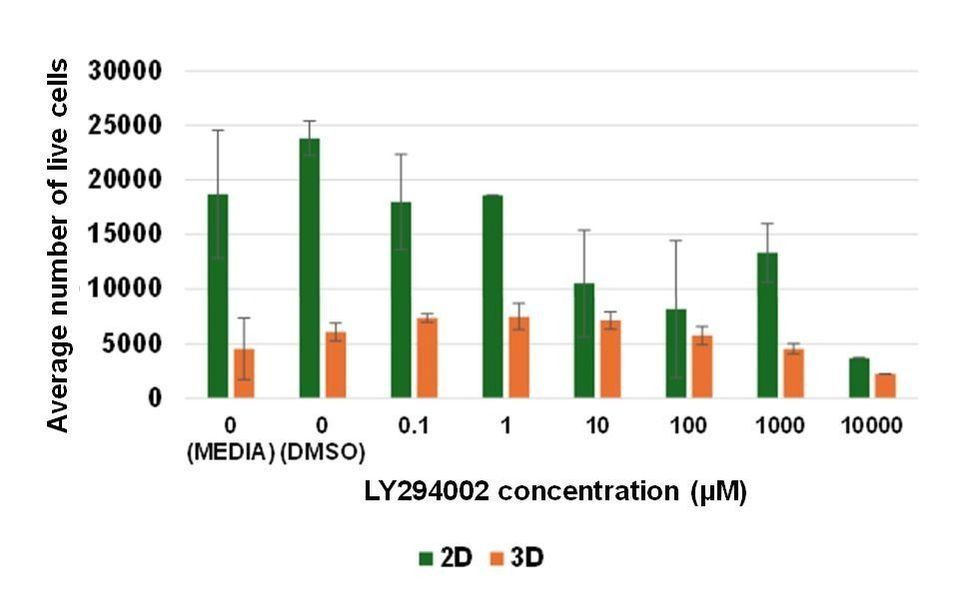
Figure 2. LY294002 inhibits HEK293 proliferation on 2D and 3D culture surfaces. HEK293 cells cultured on a 2D surface and on 3D cell microcarriers were treated with LY294002 at increasing concentrations over 24 hours. Live cells were quantified using the Alamar Blue assay. Bars on the column chart represent three technical replicates (n=3). Error bars show standard deviation between individual data points.
The live/dead assay showed that viability of cells cultured on a 2D surface was increasingly inhibited as LY294002 concentrations were elevated (Figure 3.). Areas of calcein-labelled (green) cells was measured using image analysis (ImageJ Fiji) to quantify the reduction in levels of viable cells. These data showed incremental decreases in levels of live cells as LY294002 concentrations were increased (Figure 4.), and they support the Alamar blue assay results that also showed that LY294002 caused a dose-dependent decrease in numbers of live cells on 2D culture surfaces.
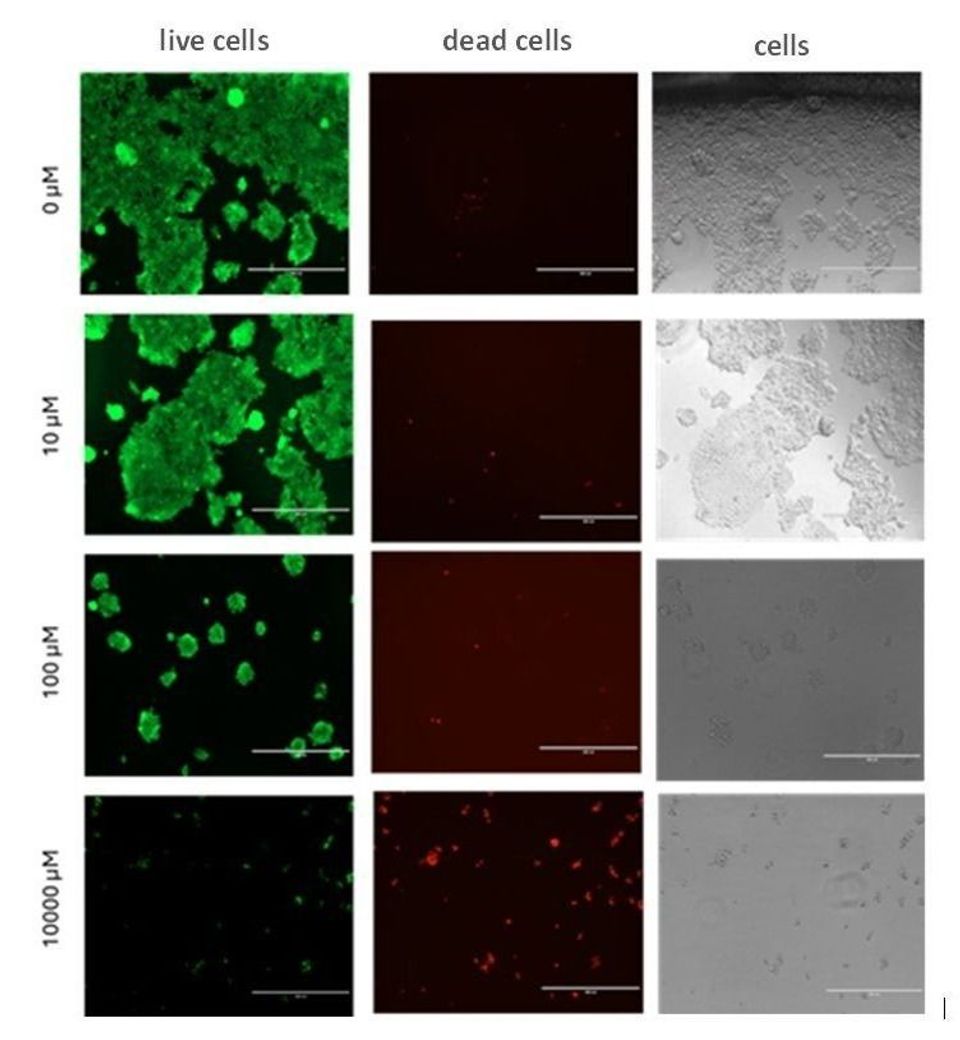
Figure 3. LY294002 inhibits viability of HEK293 cells on a 2D surface in a dose dependent manner. HEK293 cells cultured on a 2D surface were treated with LY294002 at increasing concentrations over 24 hours. Images of calcein-stained live cells (green), ethidium homodimer-stained dead cells (red), and phase contrast images of cells (grey) were acquired at 10X magnification. Images represent one experimental replicate (n=1).
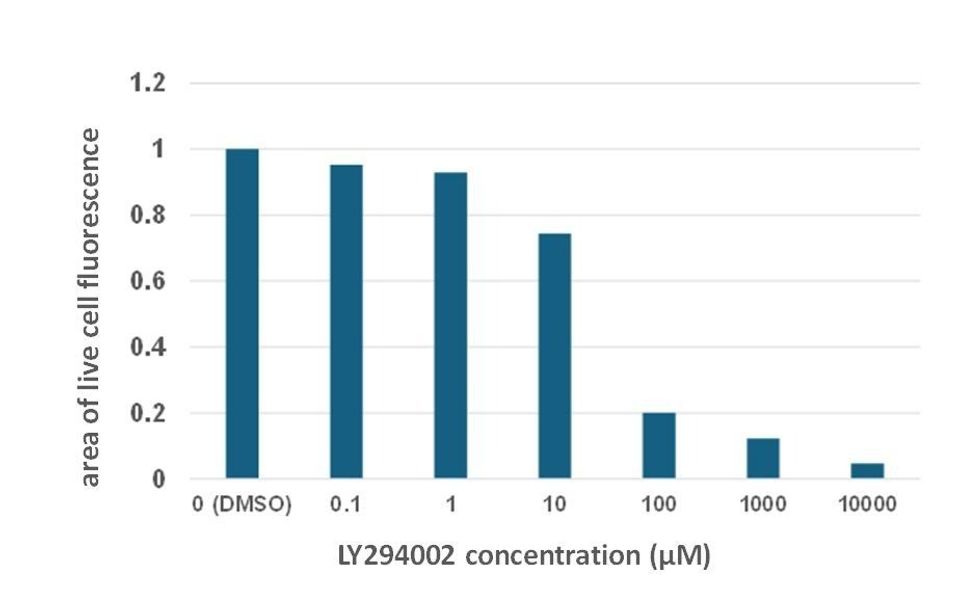
Figure 4. Quantification of live cells from live/dead assay images shows that LY294002 inhibits HEK293 cell viability in a dose dependent manner under 2D culture conditions. HEK293 cells cultured on a 2D surface were treated with LY294002 at various concentrations over 24 hours. ImageJ Fiji was used to quantify the area of live cell fluorescence in the presence of the drug vehicle (1% (v/v) DMSO) or increasing concentrations of LY294002. Bars on the chart represent one experimental replicate (n=1).
Limitations of the study include lack of experimental replicates that would indicate reproducibility of the work, and the loss of cell microcarriers due to medium removal was not measured. The great difference between levels of live cells quantified on 2D and 3D surfaces could have been caused by loss of cell loaded microcarriers during medium changes. Tumour organoids may be a more suitable in vitro model than cell microcarriers for evaluating the physiological cytotoxic effects of LY294002. Images of cells stained using the live/dead assay were acquired from only one field of view not the total areas on the well plate that contained stained cells. Therefore, quantification results from image analysis of stained cells are not accurate measurements of viable and non-viable cell levels.

Another issue was that cells were cultured on 2D and 3D surfaces for only 24 hours which may not have been a sufficient period of time to allow them to attach securely. This may be addressed by designing a time course experiment to examine cell attachment to 2D surfaces and 3D matrices. Overall, results from the present study show that LY294002 inhibits tumour cell viability in physiological 3D environments and under artificial 2D conditions but in vitro evaluation of LY294002 cytotoxicity under those conditions requires optimisation to accurately indicate efficacy of the drug. The current study may contribute to drug development pipelines by supporting the design of physiological in vitro small molecule drug testing platforms that can be translated to in vivo studies.
References
(1) Jiang, H., Fan, D., Zhou, G., Li, X. and Deng, H. (2010). Phosphatidylinositol 3-kinase inhibitor (LY294002) induces apoptosis of human nasopharyngeal carcinoma in vitro and in vivo. Journal of Experimental & Clinical Cancer Research, 29(1).
I have gained a valuable insight into the operation and organisation of a research facility, as well as many useful research skills. As I cultured my cells, I improved my aseptic technique, and analysis of my cell activity data allowed me to achieve a good understanding of how to design experiments to examine effects of a small molecule drug on cell activity. I used a fluorescence microscope for the first time. I contributed to the design of the experiment, wrote the main part of the lab report independently, applied lab safety protocols, and performed a number of different assays that I had not worked on previously, to acquire my results.
If you would like to know more about RESILIENCE courses, placements and outreach events, please
