Training Courses
 The RESILIENCE partners offer a range of training courses and outreach events across the UK and throughout the year. Most courses are run to regularly updated advertised dates but we also welcome enquiries about in-house provision of customised training which can be provided at a date and place to match your needs. Self-paced Online Training opportunities can be found here.
The RESILIENCE partners offer a range of training courses and outreach events across the UK and throughout the year. Most courses are run to regularly updated advertised dates but we also welcome enquiries about in-house provision of customised training which can be provided at a date and place to match your needs. Self-paced Online Training opportunities can be found here.
You can filter the course list below to find a course by clicking on a Category or Tag button of interest to you.
Training Courses available from RESILIENCE
Applied Skills in Mammalian Cell Bioprocessing

This hands-on course teaches laboratory scientists and engineers how to handle and maintain mammalian cells applying appropriate aseptic techniques as well as how to evaluate cell quality using a range of analytical assays.
Data Analytics 1: Introduction to Statistics

Uncover the fundamentals of statistics, with a focus on practically implementing the methods as well as understanding why they work
Downstream Processing: Primary Recovery
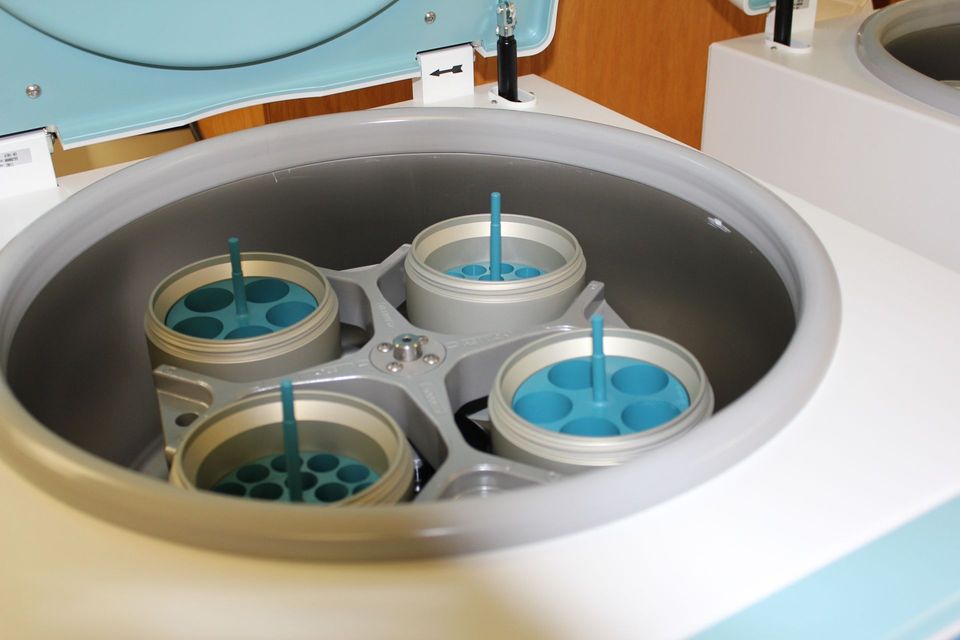
Learn how to select the best recovery options for your bioprocess by understanding the principles of all the major recovery operations: centrifugation, membrane systems, filtration, cell disruption, precipitation, flocculation. A four-day MBI module from UCL.
Fundamentals of GMP and Quality

GMP (good manufacturing practice) is the important element of Quality management to ensure medicines manufactured are of highest quality that meets the required regulatory standards. This 2-day in person course is for anyone with an interest in working in Quality in GMP Industry including QC, QA and manufacturing personnel, graduates or research scientists looking to establish their career in GMP/ ATMP industry.
Mammalian Cell Culture and Analysis (Hands-on Course)

Gain hands-on experience in the set-up and aseptic operation of single-use bioreactors for the culture of mammalian cells.
Quality by Design for Effective Bioprocess Characterisation and Validation

Get expert guidance on choosing on how best to integrate QbD, DoE and PAT into lifecycle approach to process characterisation and operation of lab-scale microbial bioreactors. 4-days of training featuring a series of stimulating interactive lectures from experts in the field and teamwork-based activities.
